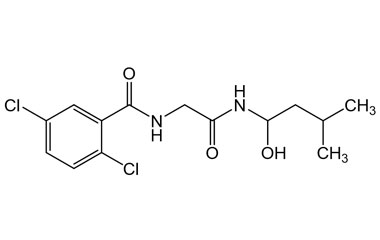
ixazomib and its Impurities
Ixazomib is used in combination with lenalidomide and dexamethasone for the treatment of multiple myeloma in adults after at least one prior therapy. It is taken by mouth in form of capsules. Reference standards of Ixazomib API,and its pharmacopeial, non pharmacopeial impurities, and stable isotopes are listed below.

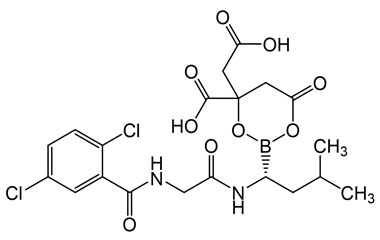
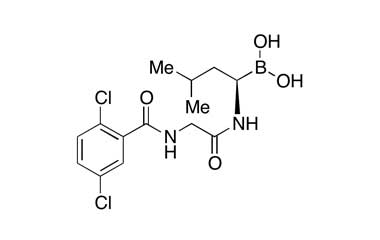
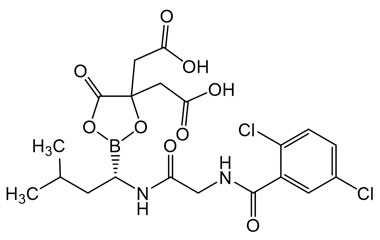
4-(Carboxymethyl)-2-((R)-1-(2-(2,5-dichlorobenzamido)acetamido)-3-methylbutyl)-6-oxo-1,3,2-dioxaborinane-4-carboxylic Acid
4-(Carboxymethyl)-2-((R)-1-(2-(2,5-dichlorobenzamido)acetamido)-3-methylbutyl)-6-oxo-1,3,2-dioxaborinane-4-carboxylic Acid
Catalogue No.:PA 09 0551000
CAS :
1201902-80-8
Molecular Formula : C20H23BCl2N2O9
Molecular Weight : 517.12

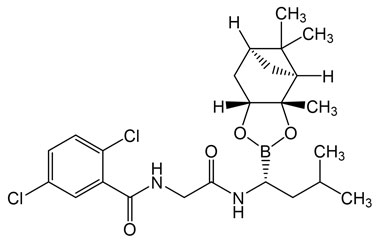
2,5-Dichloro-N-(2-(((R)-3-methyl-1-((3aS,4S,6S,7aR)-3a,5,5-trimethylhexahydro-4,6-methanobenzo[d][1,3,2]dioxaborol-2-yl)butyl)amino)-2-oxoethyl)benzamide
2,5-Dichloro-N-(2-(((R)-3-methyl-1-((3aS,4S,6S,7aR)-3a,5,5-trimethylhexahydro-4,6-methanobenzo[d][1,3,2]dioxaborol-2-yl)butyl)amino)-2-oxoethyl)benzamide
Catalogue No.:PA 09 0551007
CAS :
1201903-02-7
Molecular Formula : C24H33BCl2N2O4
Molecular Weight : 495.25

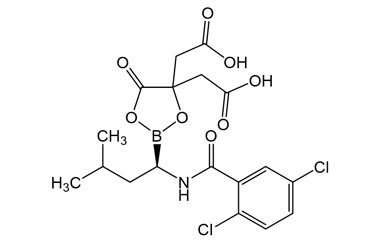
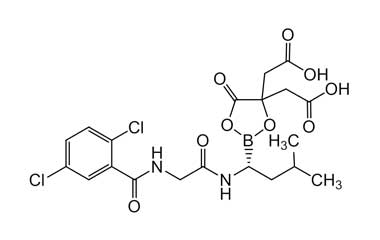
(R)-2,2'-(2-(1-(2,5-Dichlorobenzamido)-3-methylbutyl)-5-oxo-1,3,2-dioxaborolane-4,4-diyl)diacetic Acid
(R)-2,2'-(2-(1-(2,5-Dichlorobenzamido)-3-methylbutyl)-5-oxo-1,3,2-dioxaborolane-4,4-diyl)diacetic Acid
Catalogue No.:PA 09 0551009
CAS :
NA
Molecular Formula : C18H20BCl2NO8
Molecular Weight : 460.07