alendronate sodium
Fine chemicals
- Alendronic acid was first described in 1978 and approved for medical use in the United States in 1995. It is available as a generic medication. Reference standards of Alendronate Sodium API, and its pharmacopeial, non pharmacopeial impurities, and stable isotopes are listed below.
stdClass Object
(
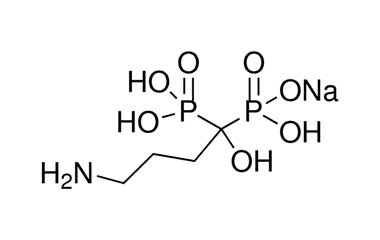
[pname] => Alendronate Sodium
[catalogue_number] => PA 54 03000
[category_ids] => ,81,78,70,82,72,
[chemical_name] =>
[weight] => 271.08
[form] => C4H12NNaO7P2
[cas] => 129318-43-0
[pslug] => 129318-43-0-alendronate-sodium-api-pa5403000
[latest_product] => 0
[linkproducts] => 1
[offers_id] =>
[offers_name] =>
[offers_status] =>
[offers_start_date] =>
[offers_end_date] =>
[pageview] =>
[offers_slug] =>
[offers_product_id] =>
[offers_product_code] =>
[offers_master_id] =>
[offer_percentage] =>
[offers_product_main_cat] =>
)
Catalogue No.:PA 54 03000
Molecular Formula : C4H12NNaO7P2
Molecular Weight : 271.08
stdClass Object
(
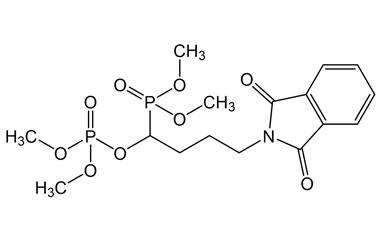
[pname] => 1-(Dimethoxyphosphoryl)-4-(1,3-dioxoisoindolin-2-yl)butyl dimethyl Phosphate
[catalogue_number] => PA 54 0031000
[category_ids] => ,78,70,82,72,
[chemical_name] =>
[weight] => 435.31
[form] => C16H23NO9P2
[cas] => NA
[pslug] => 1-dimethoxyphosphoryl-4-1-3-dioxoisoindolin-2-yl-butyl-dimethyl-phosphate-pa540031000
[latest_product] => 0
[linkproducts] => 0
[offers_id] =>
[offers_name] =>
[offers_status] =>
[offers_start_date] =>
[offers_end_date] =>
[pageview] =>
[offers_slug] =>
[offers_product_id] =>
[offers_product_code] =>
[offers_master_id] =>
[offer_percentage] =>
[offers_product_main_cat] =>
)
1-(Dimethoxyphosphoryl)-4-(1,3-dioxoisoindolin-2-yl)butyl dimethyl Phosphate
Catalogue No.:PA 54 0031000
Molecular Formula : C16H23NO9P2
Molecular Weight : 435.31