bromfenac sodium
Heterocycles
- The FDA and European approvals for Bromfenac Sodium are for use one day before and two weeks following cataract surgery for the treatment of ocular inflammation and pain. It is a nonsteroidal anti-inflammatory drug.. Reference standards of Bromfenac Sodium API, and its pharmacopeial, non pharmacopeial impurities, and stable isotopes are listed below
stdClass Object
(
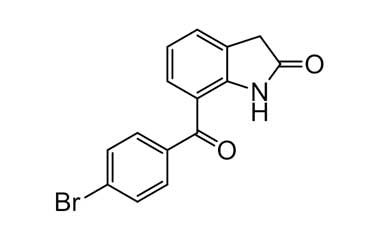
[pname] => 7-(4-Bromobenzoyl)indolin-2-one
[catalogue_number] => PA 02 47530
[category_ids] => ,100,80,71,76,75,70,78,82,
[chemical_name] =>
[weight] => 316.15
[form] => C15H10BrNO2
[cas] => 91713-91-6
[pslug] => 91713-91-6-7-4-bromobenzoyl-indolin-2-one-pa0247530
[latest_product] => 0
[linkproducts] => 0
[offers_id] =>
[offers_name] =>
[offers_status] =>
[offers_start_date] =>
[offers_end_date] =>
[pageview] =>
[offers_slug] =>
[offers_product_id] =>
[offers_product_code] =>
[offers_master_id] =>
[offer_percentage] =>
[offers_product_main_cat] =>
)
7-(4-Bromobenzoyl)indolin-2-one
Catalogue No.:PA 02 47530
Molecular Formula : C15H10BrNO2
Molecular Weight : 316.15